Preface — I have been holding off on sharing my summary of the FDA’s vaccine advisory committee (which is below the line — scroll down) because we are at one of those moments where the path forward is not clear. Global case counts are down (at the moment). But deaths are increasing. Some individuals who are healthy fully vaccinated individuals are living in fear — the same fear we all experienced in March of 2020. And despite vaccines, treatment, home tests, and a less virulent virus — they are so fearful of COVID. Others — who are at high risk — are asking important questions about how they should navigate the future and how the rest of us can cooperate with them so that they can remain active and healthy. Vaccine uptake needs to increase. We could use a better vaccine. Communications need to be clearer. The public needs digestible and actionable information.
A path forward is needed.
But there is no clear path.
To me, the future (not just of COVID, but of public/community health) looks something like a trainyard. A single set of tracks comes to a juncture (which is where I am imagining we are at this moment in time) and there are numerous paths forward. Some paths overlap with one another. Some paths are distinct. There are switches to move this way or that and those switches/decisions will determine the path forward. Some of those switches will lead to a track heading one way. With no room to turn back or reconnect with the other tracks. Other switches will allow for a shift this way or that, but with another switch, the direction can change or come back to another path.
But we can only see a little way down the track. What is lurking beyond the fog is unknown. There could be blue skies ahead. Or a storm. Or more fog. Or something unknown like a tree on the tracks or a blocked tunnel.
A path forward is needed. But there is no clear path.
In addition to the FDA meeting on Thursday, WHO’s emergency committee met on Friday and again on Saturday to discuss whether COVID-19 still warrants a public health emergency of international concern (PHEIC). Recent data show that cases are declining worldwide, but deaths are rising. Cases in China are declining.
WHO’s decision matters; FDA’s decision matters; what CDC has to say matters; and our ability to use all of this information to remain healthy and active and engaged matters. There are layers upon layers of information.
If COVID is no longer a PHEIC, what will it mean to public health priorities? how does that decision support or contrast the FDA's decision about vaccines? what will it mean for vaccine developers? how will it impact the public’s understanding of risk? how will it impact our individual decisions to stay healthy? will individuals and their healthcare providers continue to talk about disease transmission, illness, prevention, and treatment?
And if it is still a PHEIC — how do we continue to do the work necessary to prevent, slow, diagnose, and treat people with a disease that the world is so stinking tired of talking about? How does the PHEIC retain its integrity as we enter year 4 of the pandemic?
I don’t have answers to these questions.
I’ve been waiting for a decision from the WHO for more than 48 hours. But they are still discussing the current situation and making decisions.
What is the current situation?
We are at the start of year 4 of the pandemic.
We are at the start of year 3 of a global vaccination campaign.
We have no crystal ball to show us the future.
We need a clear path forward.
In light of these four facts about the COVID situation above, the FDA vaccine advisory committee met on Thursday. Their task was straightforward…
First, they were asked to approve a proposal to streamline the COVID-19 vaccine campaign by offering just the bivalent shot — as the primary series and booster dose.
The committee voted unanimously to move to one type of shot — the bivalent vaccine that includes the mRNA of the original SARS-CoV-2 virus + Omicron.
The move to a single type of vaccine simplifies things for providers and vaccine recipients. The move is also backed by science. Moderna shared the results of a recent study (published online on Wednesday)
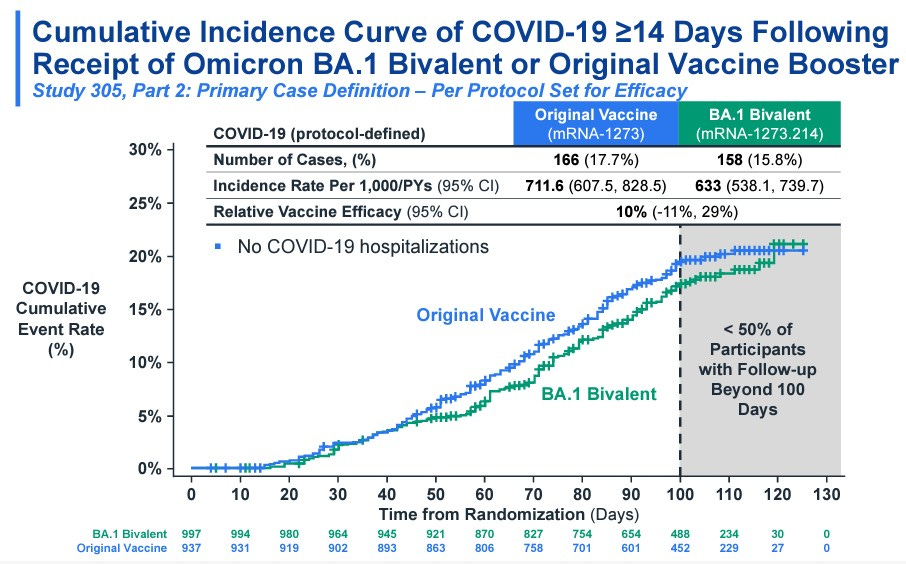
The second thing the committee was asked to do on Thursday was to discuss (no vote) whether or not they were supportive of a plan to move to a single annual COVID shot — that would be matched annually to circulating strains of the virus.
Moving to an annual shot — similar to our annual flu shot — simplifies vaccine communications and education. But there are a lot of uncertainties. Specifically, the committee debated whether or not COVID has established a seasonal pattern. The question that was raised over and over again was if you can only get one shot per year, when do you get it in order to get maximum benefit?
No clear answer was provided.
There were also conversations about the fact that the move was one of convenience/simplicity, but not backed by scientific evidence.
Do we simplify the process in order to get some benefit? or do we keep things a little complicated for providers and the public for a little while longer until we have the data to provide an evidence-based approach?
There is no simple answer to this question.
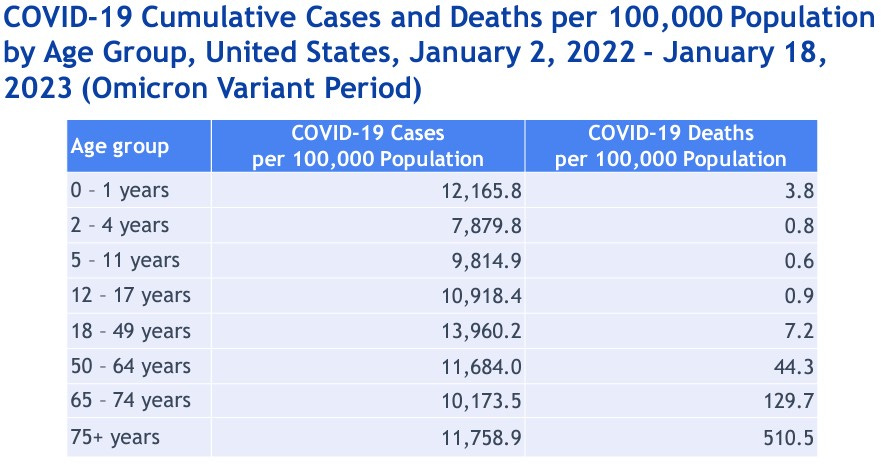
Other questions that were raised included whether or not everyone needs to get an annual shot. Data from CDC show that severe disease, hospitalizations, and deaths are occurring most often among the elderly and very young babies, who have not been exposed to COVID.
The committee also pointed out that the use of only antibody titers as a marker for immune response may be shortsighted. Maybe it is time to look for new measures and markers of immune response and broaden our thinking and definitions of vaccine effectiveness. Dr. Stanley Perlman, who chairs the FDA vaccine advisory committee stated —
“I wonder if using neutralizing antibodies as the sole correlate of protection, which is what we kind of do, needs to be upgraded or changed because it seems like people are still pretty well-protected, even though the antibody titers are variable.”
He was referring to data shared on Thursday that show that individuals who have been vaccinated months ago are not landing in the hospital, suffering from severe disease, and dying.
So many questions. Many pros/cons related to potential paths forward.
What did the committee decide regarding an annual COVID shot?
Officially, nothing. After hours of discussion, it appears that the committee is supportive of a plan to move to a single annual shot, but the actual decision will be made at a later date. The can has been kicked down the road — so to speak.
Where does CDC fit into these decisions?
Now that FDA has made the decision about the vaccine, CDC’s Advisory Committee on Immunization Practices must meet and approve the decision. The committee is scheduled to meet in late February, but the COVID vaccine is not on the agenda. We are expecting that they will convene an additional meeting in early February to discuss the move to a single vaccine type and annual COVID shots. Stay tuned…
Do you have further questions?




