J&J Vaccine -- The Pause Is Lifted
Advisory Committee on Immunization Practices recommends J&J vaccine for all
Eleven days ago, FDA and CDC announced a pause in the distribution and administration of the J&J vaccine. Following that announcement, the Advisory Committee on Immunizations Practices (ACIP) met on April 14 to discuss a serious adverse side effect — CVST with thrombocytopenia (low platelet counts) — associated with the J&J vaccine.
TODAY the ACIP met again to discuss the risks and benefits of the vaccine and determine/vote if the vaccine should be used in the United States.
Details of the ACIP meeting are below. In short, the ACIP voted (10 to 4 with one person abstaining) to recommend the J&J vaccine for everyone 18 years of age and older. The J&J vaccine is safe for the vast majority of people and is saving lives. The J&J vaccine will come with a warning. Dr. Joanne Waldstreicher, chief medical officer for Johnson & Johnson, shared that the warning will include language along the lines of —
“Recipients of the shot should be instructed to seek immediate medical attention if they develop shortness of breath, chest pain, leg swelling, persistent abdominal pain," as well as neurological symptoms, such as blurred vision or severe and ongoing headaches.”
According to Helen Branswell at STAT news, women under 50 have permission to get the J&J vaccine if they are comfortable with the risks associated with the vaccine.
According to data presented at today’s meeting, 15 women have experienced this adverse side effect associated with the J&J vaccine. The highest rates of CVST with low platelet counts were seen among women aged 30-39 (rate is 11.8 cases per million individuals). And there were no cases among women 65 and older.

Much of today’s conversation focused on the risks posed by the J&J vaccine versus the benefits provided by a one-dose vaccine that does not need to be stored in a deep freezer. The data below models what would happen in the J&J vaccine were made available tomorrow…
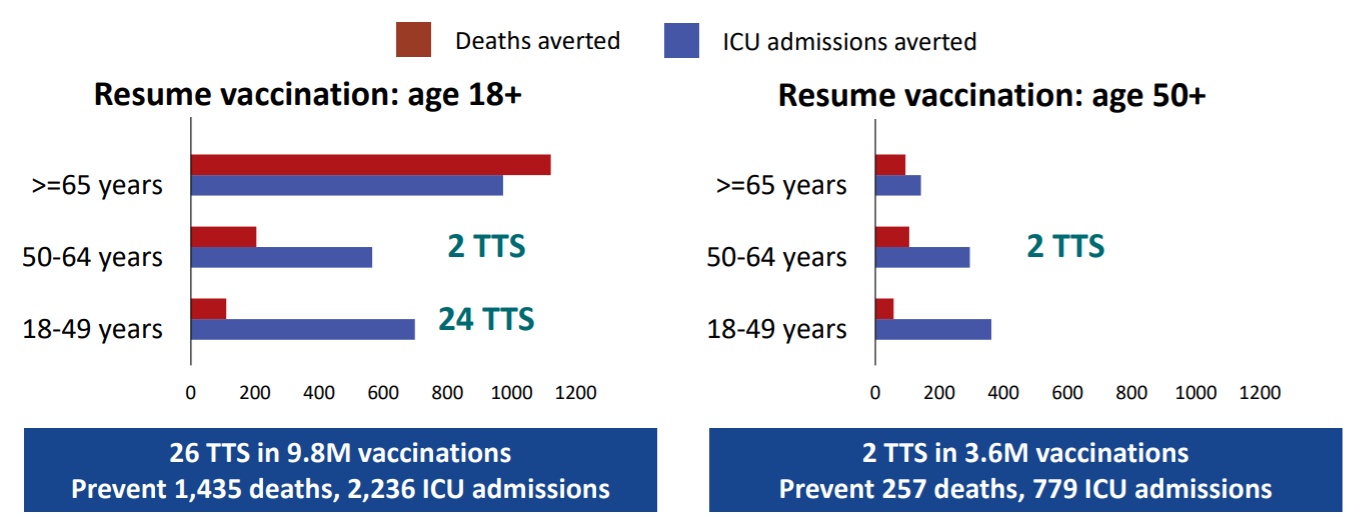
What we see here is that if the J&J vaccine is made available to all adults 18 years of age and older — we anticipate another 26 CVSTs with low platelets if 9.8 million doses of the J&J vaccine are administered. We also anticipate with the same number of doses more than 1400 deaths and 2200 ICU admissions will be prevented.
If on the other hand, the J&J vaccine were available to only individuals 50 years of age and older, there would be only two (anticipated) cases of CVSTs with low platelet counts, but only 237 deaths and 779 ICU admissions would be prevented.
When the ACIP looked at this data, they believed the benefits of the J&J vaccine outweigh the risks posed by this rare adverse side effect.
Additionally, the J&J vaccine (which is a single dose and can be stored in a refrigerator) provides public health professionals and clinicians the ability to vaccinate hard to reach populations, including those who live in rural/remote communities and those who are homebound, homeless, transient (e.g., migrant workers), and incarcerated.
The J&J vaccine will be available with the FDA warning in the next few hours.



